



February 2026 Crop Talk
Volume 23 | Issue 1
Published February 27, 2026
Greetings everyone,
As you get ready for the 2026 growing season, it may help to focus on the usual early-season pinch points: starting clean, staying ahead of the first flush of insects and diseases, and setting up a scouting routine that’s realistic. Using tools like good record keeping, timely scouting, and weather-based decision support can make management more predictable, and, hopefully, more profitable.
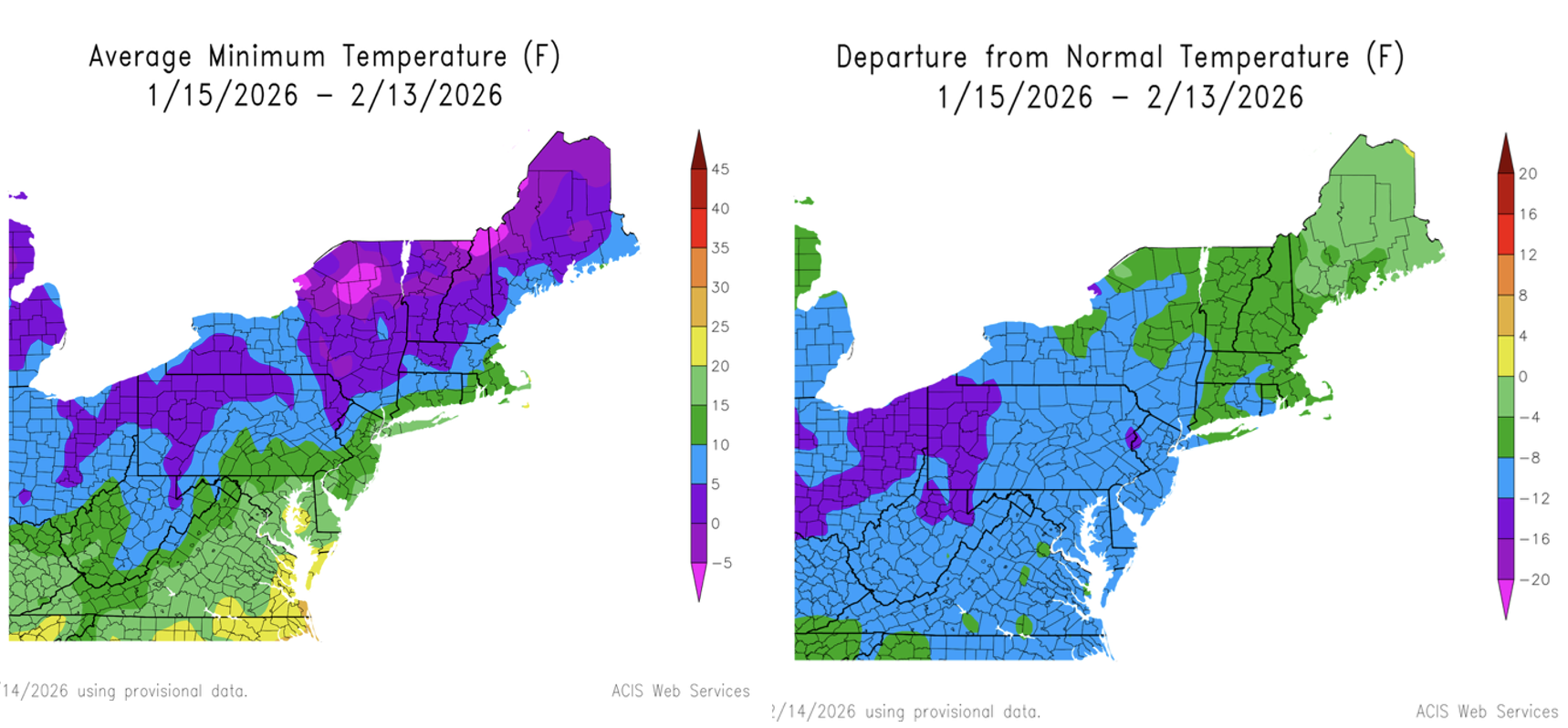
A question that came up this winter was whether a colder winter will reduce insect pests this spring/summer. The short answer is it depends a lot on the pest and where it overwinters (soil, residue, leaf litter, or structures). I’ve included an article in this Crop Talk ‘How cold is too cold for insect pests?’ to help put winter temperatures in context and what they may (and may not) mean for spring pest pressure.
A second topic that’s come up recently is soil steaming. If you’re considering it, we have a resource written by Paul Bucciaglia (Fort Hill Farm, New Milford) that walks through the soil steaming process and discusses impacts on weeds, soil microbial activity, soil nutrients, and nematodes, along with practical thoughts on the economics of the technology. Read the archived article on soil steaming.
I also want to highlight our vegetable factsheets, which we are actively revising to serve as a quick reference as you head into the season. So far, we’ve updated fact sheets related to tomatoes and pumpkin diseases, and we’ll continue adding more in 2026. You can find the fact sheets on our website.
Finally, in this issue of Crop Talk you will find details on upcoming webinars, including sessions on farm profitability and how to use weather tools to improve IPM timing (degree days, weather summaries, and disease risk tools).
As always, please reach out if there’s something you want help thinking through as the season approaches, cropping plans, pest concerns, fertility questions, setting up scouting, or anything else. And if there are topics you’d like us to cover in 2026 through newsletters or meetings, please let us know. We’re always looking for ways to make our programs and resources more useful to you.
Best wishes for a successful 2026 season!
Thanks,
Shuresh Ghimire
Extension Vegetable Specialist
shuresh.ghimire@uconn.edu

In This Issue
- How Cold is Too Cold for Vegetable Insect Pests
- Get a Head Start on Herbs this Spring
- Spring is the time for Tiphia and Peonies
- Using Cornell NEWA for Vegetable IPM Decisions
- Rosy Apple Aphid (Dysaphis plantaginea)
- What a Caterpillar Tunnel (and Climate Smart Microgrants) Can Do
- Growing in the City-Season Extension Without a High Tunnel
- UVM Food Systems Research Institute: Earn $50 for Short Survey!
- Upcoming Events
- Quick Links to IPM Resources

How Cold is Too Cold for Vegetable Insect Pests?
By: Shuresh Ghimire, Vegetable Extension Specialist, UConn Extension
This winter, Connecticut has had colder than historical average temperatures (-4 to -12 departure; Figure 1). A colder-than-normal winter can reduce certain insect pest populations, but winter kill is highly pest-specific and depends on (a) which life stage overwinters, (b) where that stage hides (soil vs. residue vs. buildings/woodlots), and (c) how long it experiences damaging temperatures.
A key concept behind “kill temperatures” is how insects respond to freezing. Insects are broadly classified as either freeze-intolerant (freeze-avoidant) or freeze-tolerant. Freeze-intolerant insects cannot survive the formation of internal ice; if their body fluids freeze, they die. These species survive winter by lowering their supercooling point (SCP), the temperature at which spontaneous freezing occurs, and by producing cryoprotectants or seeking insulated microhabitats. For these insects, the SCP serves as a practical “freeze-kill” benchmark (Sheffield, 1998). Many major vegetable pests fall into this category, including Colorado potato beetle adults, cabbage maggot pupae, onion maggot pupae, and brown marmorated stink bug adults, all of which rely on supercooling and are killed if freezing occurs. In contrast, freeze-tolerant insects can survive controlled internal ice formation by stabilizing tissues and regulating where ice develops; this strategy is less common among major vegetable pests but has been documented in some overwintering Lepidoptera, including European corn borer (Grubor-Lajšić et al., 1992). Because most economically important vegetable insect pests are freeze-intolerant, winter mortality depends not only on how low temperatures drop relative to their SCP, but also on how long those temperatures persist and whether the insects are buffered by soil, residue, litter, or structures.
This same “cold exposure” framework also applies to beneficial insects: a colder winter can reduce survival of pests, but it can also reduce survival of pollinators and natural enemies, while a warmer winter may allow greater carryover of both pests and beneficials.
Sometimes minimum air temperature can be misleading. Most overwintering stages are not sitting in free air. Soil temperatures, especially a few inches down, are buffered. Crop residue/leaf litter can insulate and reduce exposure. Buildings, bark crevices, stone piles, hedgerows can dramatically moderate cold exposure. Snow cover often increases insect survival by insulating soil and litter. Buffered habitats such as hedgerows, field margins, and leaf litter can act as refuges for overwintering pests and beneficial insects alike, so the net outcome in spring can reflect not just pest survival, but also how well biological control agents and pollinators survived the winter.
In addition, large structures (heated buildings, barns, and even unheated high tunnels warmed by sunlight) can create “warm edge zones” outdoors: the thermal mass of the structure and solar heating can keep adjacent soil and crop debris warmer for several feet, not just inside the structure. In practice, this can lead to higher-than-expected early-season pest densities right next to structures (for example, along high tunnel edges) and occasionally earlier-than-expected appearances of pests that typically overwinter poorly. This effect tends to be most noticeable on smaller and urban farms, where a greater proportion of the farm area is influenced by buildings, tunnels, paved surfaces, windbreaks, and other heat- and shelter-modifying features.
So, a “colder winter” can reduce populations of some pests, but it doesn’t guarantee low spring pressure- especially for pests protected by soil or structures (Table 1).
Pest (key crops) |
Temperature (°F) for activity |
Published lethal cold benchmark (°F) |
Overwinter life stage (typical) |
Practical winter-kill interpretation |
| Cabbage maggot (Delia radicum) (brassicas) |
Activity starts when the soil temperature exceeds 40. | Pupae show a strong SCP peak near −9. (Soil temperatures exceeding 95 in the top 2-3 inches will kill eggs.) (Košťál & Šimek, 1995) | Pupa in soil | Because pupae are in soil, air temps must be extremely low and soil must freeze deeply to approach these lethal benchmarks. Snow cover often protects them even if air temp is -9. |
| Onion maggot (Delia antiqua) (alliums) |
Activity starts when the soil temperature exceeds 40. | Puparia SCP naturally drops to about −16.6. Soil temperatures above 95 for several consecutive days will kill eggs (Kayukawa & Ishikawa, 2009) | Pupa in soil | Extremely cold‑hardy as pupae. |
| Seedcorn maggot (Delia platura) (seedlings: brassicas, corn, beans, cucurbits, etc.) |
Flight activity reported from 39–84, with maximum near 61. | Not standardized | Pupa in soil | Treat winter kill as unreliable for decision-making. Spring risk is often driven more by cool, wet soils + decaying organic matter/manure/cover crop residues than by winter minimums. |
| Flea beetles (crucifer flea beetle, striped flea beetle) (Phyllotreta cruciferae, P. striolata) (brassicas) |
Adults emerge from overwintering sites in soil or leaf litter when temperatures reach approximately 57. | SCP at 13 to -11 (Hiiesaar et al., 2012) | Adult in leaf litter/field edges | Even with cold winters, assume early feeding risk on seedlings and protect the cotyledon–1st true leaf window; winter kill is too variable to bank on. |
| Striped cucumber beetle (Acalymma vittatum) (cucurbits) |
Adults emerge and begin host-seeking when temps reach 54–62 | Not standardized | Adult in debris/edges/woodlots) | Winter kill can occur, but survival is strongly driven by shelter quality (leaf litter/edges). Plan for early pressure unless you have strong local evidence otherwise. |
| Squash vine borer (Melittia cucurbitae) (cucurbits) |
Primarily a warm-season pest (adult flight mid‑season; benchmark varies regionally) | Not standardized | Larva in soil cocoon (overwinters; pupates the following summer) | Soil overwintering makes winter kill hard to predict from air temps. Cultural tactics (crop rotation; destroying vines; tillage that disrupts cocoons) are often more dependable than hoping for winter mortality. |
| Colorado potato beetle (Leptinotarsa decemlineata) (potato/eggplant; sometimes tomato) |
Overwintering adults survived better when soil temps stayed near 32 (soil buffering) | Mean SCP for overwintering adults reported near 21 (Hiiesaar et al., 2014) | Adult in soil burrow | Deep soil buffering often prevents exposure to lethal temps. Winter kill is most plausible with shallow overwintering + deep frost + low snow cover. |
| European corn borer (Ostrinia nubilalis) (sweet corn; peppers) |
Development begins when temperatures exceed 50 | Lower lethal temperatures reported around −18.4 (Grubor-Lajšić et al., 1992) | Larva in corn stalks/residue | This pest is very winter-hardy. A cold winter usually does not eliminate it. Residue management (shredding/burying) often matters more than winter minima. |
| Tarnished plant bug (Lygus lineolaris) (many crops: strawberries, leafy greens, brassicas, beans, etc.) |
Emerge in spring, when temperatures reach 46-50. | Do not experience significant mortality until temperatures drop below 14 (Canola Council of Canada, 2024). | Adult (sheltered litter/edges) | Cold snaps can reduce populations if overwinter microhabitats approach SCP conditions. But adults actively choose protected sites; winter kill is variable. |
| Brown marmorated stink bug (Halyomorpha halys) (many veg: sweet corn, peppers, tomatoes, beans) |
Nymphs and eggs require temperatures above
53-59 to develop |
Exposure-trial benchmarks (diapausing adults) below 13 to 18 (Scaccini et al., 2020) | Adult (often in structures or sheltered natural refuges) | A cold winter can cause meaningful mortality if overwintering sites cool enough. Buildings and deep shelter can greatly buffer temps, so outcomes vary farm-to-farm. |
So, what does a 5- 15°F minimum (Jan 13–Feb 13, 2026) actually mean for winter-kill?
For many of the vegetable pests in this article, the key point is that they overwinter in protected sites (soil, residue, leaf litter, crop debris, or buildings). Those shelters often stay warmer than the coldest air temperature, especially a few inches into soil or under snow/residue. So, the 5–15°F “official” minimum does not automatically translate into widespread mortality. Just as important, overwintering survival is not uniform across a farm, even within the same field. Some individuals are well sheltered (deeper soil, thicker litter, protected edges, near hedgerows, or adjacent to buildings/high tunnels), while others are exposed and die. As a result, cold winters more often change the proportion of each pest population that survives, rather than producing an “all-or-nothing” outcome where one species survives and another does not. This means that even cold-hardy pests can show noticeable drops in early-season pressure after a colder-than-usual winter, because a subset of the population was not sufficiently protected.
That said, the 5–15°F minimum is meaningful for a few pests, particularly those that overwinter as adults in marginal shelters or those with published cold-injury benchmarks near that range:
- Brown marmorated stink bug (BMSB): Mortality can occur in the low teens. A 5-15°F minimum could therefore reduce populations in poorly insulated overwintering sites (e.g., cracks in woodpiles, exposed sheds, thin-barked trees). However, adults in heated buildings or well-buffered structures may be largely protected.
- Striped cucumber beetle (and other exposed-overwintering adults): Free air temperatures of 5-15°F could be lethal if beetles are directly exposed. But beetles typically overwinter under leaf litter and in protected edges, so outcomes depend heavily on how insulated those overwintering sites were (snow cover, deep litter).
For the soil-overwintering pests discussed above, cabbage maggot, onion maggot, squash vine borer, and Colorado potato beetle: air minimums of 5-15°F are usually a weak predictor by themselves. Soil a few inches down often remains warmer than the coldest air, so meaningful winter kill requires deep frost penetration and limited insulation. Similarly, for pests like European corn borer (overwintering larvae in crop residue), cold tolerance is generally high enough that 5-15°F air minimums are unlikely kill the populations significantly; residue management often matters more than winter minima. The 5-15°F minimum suggests the best chance of noticeable winter-related reduction is in pests that overwinter as exposed adults like BMSB and striped cucumber beetles (especially where shelters were poor). For most soil- or debris-overwintering pests, expect variable effects and do not assume low spring pressure. It is safest to plan for typical spring pressure and use scouting, traps, and degree-day timing to confirm what survived, especially in localized “warm pockets” near structures and sheltered field edges where survivors may concentrate.
Acknowledgements
I would like to thank Alireza Shokoohi, UMass Extension, for his review and feedback on this article.
References
Canola Council of Canada. (2024). Lygus bugs. Canola Encyclopedia. https://www.canolacouncil.org/canola-encyclopedia/insects/lygus-bugs/
Hiiesaar, K., Kaasik, R., Williams, I., Kruus, E., Jõgar, K., Metspalu, L., Mänd, M., Ploomi, A., & Luik, A. (2012). Cold hardiness of horseradish flea beetle (Phyllotreta armoracia (Koch)). Žemdirbystė = Agriculture, 99, 203–208.
Hiiesaar, K., Karise, R., Williams, I., Luik, A., Metspalu, L., Jõgar, K., Eremeev, V., Ploomi, A., Kruus, E., & Mänd, M. (2014). Cold tolerance of Colorado potato beetle (Leptinotarsa decemlineata Say) adults and eggs. Žemdirbystė - Agriculture, 101(4), 431–436. https://doi.org/10.13080/z-a.2014.101.055
Jian, F. (2024). Lethal and mobile variation of stored product insects and mites under low temperatures. Journal of Stored Products Research, 105, 102240. https://doi.org/10.1016/j.jspr.2023.102240
Kayukawa, T., & Ishikawa, Y. (2009). Chaperonin contributes to cold hardiness of the onion maggot Delia antiqua through repression of depolymerization of actin at low temperatures. PLoS ONE, 4(12), e8277. https://doi.org/10.1371/journal.pone.0008277
Košťál, V., & Šimek, P. (1995). Dynamics of cold hardiness, supercooling and cryoprotectants in diapausing and non-diapausing pupae of the cabbage root fly, Delia radicum L. Journal of Insect Physiology, 41(7), 627–634. https://doi.org/10.1016/0022-1910(94)00124-Y
Scaccini, D., Vanishvili, L., Tirello, P., Maistrello, L., & colleagues. (2020). Lethal and sub-lethal effects of low-temperature exposures on Halyomorpha halys (Hemiptera: Pentatomidae) adults before and after overwintering. Scientific Reports, 10, 15231. https://doi.org/10.1038/s41598-020-72120-5
Sheffield, C. S. (1998). Overwintering biology of the tarnished plant bug, Lygus lineolaris (Palisot de Beauvois) (Hemiptera: Miridae), in Nova Scotia, and the potential use of ice nucleating active bacteria for insect pest control (Master’s thesis, Acadia University). Library and Archives Canada. https://central.bac-lac.gc.ca/.item?app=Library&id=MQ27589&oclc_number=46580717&op=pdf
Get a Head Start on Herbs this Spring
By: Charles Krasnow, Associate Extension Educator, UConn Greenhouse and Nursery

Herbs are very popular as potted crops and planted in the garden. Greenhouse operations in Connecticut sell herbs as transplants, or as potted culture to be grown until harvest on a patio, balcony, or kitchen windowsill. The popular herb crops include basil (Ocimum basilicum), rosemary (Rosmarinus officinalis), parsley (Petroselinum crispum), and chives (Allium schoenoprasum), with many other species and less common varieties also found in production. The farm fresh flavor of these culinary herbs adds pungency and spice to many dishes, giving them widespread popularity. Many consumers enjoy these plants for the ease of culture, compact size, attractiveness, and health properties. This crop note describes some of the common diseases of herbs to watch for this spring.

Growers of herbs often direct seed their own crop or stick cuttings for propagation. Treating seed prior to planting can limit pathogens that are carried on the seed. Hot water treatment is a simple and effective practice for many herbs (excluding basil). Purchasing seed from certified suppliers can ensure sanitation practices have been followed and is a recommended practice. Common pathogens that infect during seedling production include damping off organisms, Alternaria, Botrytis, and Pseudomonas the cause of bacterial leaf spot. Alternaria leafspot and stem blight is very common with seed propagated herbs. This pathogen is commonly associated with flowers during seed production in dry climates, and can readily contaminate the seeds during processing. Production of cuttings of herbs such as rosemary can also be a challenge. Stock plants should be maintained in their own growing area, and maintained under strict sanitation conditions to limit the chances of disease spread. Pathogens such as Botrytis produce abundant spores on decaying leaves and plant debris in the greenhouse. When removing cuttings, spores of the pathogen can become airborne and stick to the cuttings, providing a means of spread in the next planting.

Root rot pathogens can also limit healthy growth and success of the herb crop. Pythium root rot is very common, and has been observed on basil, mint, lavender, and others. This pathogen is a water-mold, highly favored by wet soil. Usually seedlings are affected in trays, and after transplanting, stunting and yellowing can be observed. The symptoms may appear similar to a nutrient deficiency, when in fact Pythium is preventing normal root growth. This pathogen is especially difficult to control in greenhouses using ebb and flood benchtops with recirculating irrigation. Even a small number of infected plants can release spores that contaminate the water in the holding tanks and other nearby plants. The best method of control is limiting overwatering.
Certain fungicides are highly effective against the Pythium pathogen, but may not be permitted for use on herbs. Be sure to check the label and the UConn IPM greenhouse website for additional recommendations if you suspect Pythium has been a problem in your greenhouse.
Downy mildew is another pathogen that has become a huge concern in greenhouse herb production. Unlike the previous pathogens, downy mildew is crop specific. Notable examples include basil downy mildew, which took growers and researchers by surprise by its ability to rapidly destroy a crop. Downy mildew is also a problem on chives and some other minor herbs. Identification of the pathogen is straightforward but requires a trained eye and hand lens. Symptoms on the upper side of the leaf are usually yellowing and necrosis, Leaves that are flipped over reveal fuzzy sporulation on the underside of the leaf. The sporulation is often gray or white, but with basil it is black. Under a microscope, the spores appear like tiny grape-like clusters. To limit damage from this pathogen, fungicides and biocontrols should be applied starting at the seedling stage. Resistant basil cultivars are available that can be grown if downy mildew has been a recurring problem.
Spring is the Time for Tiphia and Peonies
By: Ana Legrand, Department of Plant Science & Landscape Architecture, University of Connecticut
If you have dealt with Japanese and oriental beetle grubs, you might have wondered what natural enemies there are to give these pests trouble. One of them is the spring Tiphia (Tiphia vernalis) parasitic wasp (Fig. 1). These shiny black wasps are solitary. They do not live in colonies or make nests, and they only have one generation per year. If you did not look for them actively, you likely would not even notice them as they are not aggressive or bothersome to people. Male wasps appear 3 to 4 days before females emerge. In Connecticut, spring Tiphia are active from the first week of May to the beginning of June with a peak in numbers observed around the last week of May. After mating, female wasps discretely burrow into the soil and hunt for grub hosts. When a host is found, the wasp stings it thus paralyzing it momentarily so she can attach one egg on the ventral groove between the third thoracic and first abdominal segments (Fig. 2). The egg stage lasts for 9-10 days, and the larval stage lasts about 20 days. The parasitic larva attaches outside the host and feeds on it until the host dies (Fig. 3). At this point, the full-grown larva spins a papery, water resistant, silken cocoon (Fig.4) and within the cocoon it transforms into the adult wasp. The cocoon not only houses the amazing transformation, but it also protects the adult wasp through the winter. The Tiphia larva and its host move down deep in the soil before Tiphia takes the last bite. Indeed, the only part Tiphia leaves of the grub is the hard head capsule. Adult Tiphia emerge in spring to start the cycle all over again.

Spring Tiphia females live for about a month and may lay 40-50 eggs on as many different grub hosts. The female wasps seek out the fully grown Japanese or oriental beetle grubs in the period when the grubs are feeding before pupation. Our surveys found spring Tiphia present in every Connecticut county, in most of Massachusetts counties and even few southern locations in New Hampshire. In one survey we found a range of 61 to 100% parasitism on the Japanese beetle grubs. For oriental beetles, we documented parasitism rates ranging from 7% to 33% in low-density grub populations.
One important question to ask is how to conserve these naturally occurring wasps to benefit the most from them.
The spring Tiphia uses nectar and insect honeydew to supplement their diet. Often these wasps are observed on maple, cherry and Tulip tree foliage looking for aphids as a source of honeydew. However, it would be best to provide them with more dependable sources of carbohydrates. By providing nectar resources to the wasp, we can enhance their survival and egg production. Extrafloral nectar from peonies significantly attracts them resulting in grub parasitism increasing near the peonies.
In our studies we have documented that peony cultivars like ‘Big Ben’, ‘Festiva’ and ‘Bowl of Beauty’ are a reliable source of nectar for these wasps. They feed on the extrafloral nectar secreted by the unopened peony flower bud and one can easily see them gathering around the buds. Again, these wasps are not aggressive and do not damage the plants as they feed on the nectar. Once the flower opens, the extrafloral nectar presence stops and Tiphia move on. Peonies make excellent cut flowers with delightful fragrance and colors. Mature plants are prolific flower producers, and these cut flowers are popular with high market demand. Now, you can enjoy growing them even more knowing that they help a natural enemy of Japanese beetle grubs.



References
Legrand, A. 2009. Evaluation of landscape ornamental plants as nectar plants for Tiphia vernalis and as host plants for pest scarab beetles. p. 73-76. In K. Guillard (ed), 2009 Annual Turfgrass Research Report, College of Agriculture and Natural Resources, University of Connecticut.
Legrand, A. 2015. Use of peonies to conserve spring Tiphia parasitoids of white grubs: a three-year study. 2014 Annual UConn Turfgrass Research Report. Department of Plant Science and Landscape Architecture, University of Connecticut.
Ramoutar, D. and A. Legrand. 2007. Survey of Tiphia vernalis (Hymenoptera:Tiphiidae) a parasitoid wasp of Popillia japonica (Coleoptera: Scarabaeidae) in Connecticut. Florida Entomologist 90(4): 780-2.
Rogers, M.E. and D. A. Potter. 2004. Potential for sugar sprays and flowering plants to increase parasitism of white grubs (Coleoptera:Scarabaeidae) by Tiphiid wasps (Hymenoptera:Tiphiidae). Environ. Entomol. 33: 619-626.
Using Cornell NEWA for Vegetable IPM Decisions
By: Shuresh Ghimire, Vegetable Extension Specialist, and Nicole Davidow, Outreach Assistant, UConn Extension
NEWA (Network for Environment and Weather Applications) is a Cornell-led web platform that turns weather data into practical decision support for scouting, spray timing, and risk forecasting. For vegetable growers, NEWA can be a helpful tool for anticipating pest and disease risk by tracking degree days, recognizing weather patterns that favor infection, and aligning management actions with crop stage and field conditions. NEWA does not replace scouting. Instead, it is one more tool in the IPM toolbox that supports better-informed decisions: when to scout more regularly, when protection is most likely needed, and when it is reasonable to hold.
Part I. Getting NEWA Set Up
Start by selecting the weather location that best represents your farm. In most cases, this will be the closest station or grid point, but the most useful choice is the one that best matches your field’s drying pattern and rainfall timing.
Once your weather location is set, treat NEWA outputs as a decision guide:
- Low risk: maintain normal scouting.
- Rising risk: scout sooner, prepare protection, and plan product rotation.
- High risk: protect susceptible blocks and avoid stretching intervals.
- After rain or overhead irrigation: reassess quickly, because risk can change fast.
Part II. Weather Summary Tools
All Weather Data Query (Weather Summary Tool)
The All Weather Data Query tool lets you view or download weather information as tables or graphs. It can provide hourly data, daily summaries, and sometimes degree-day summaries depending on the station and options selected. This tool is especially useful for understanding what actually happened in your area, particularly during patchy storm events or periods when diseases suddenly increase.
Growers can use this tool to confirm rain totals and timing, identify long wet or humid stretches that drive foliar disease, document weather for records, and find workable field windows for spraying, harvest, or cultivation.
To use it, open the All Weather Data Query tool and select a weather location (station or grid point). Then choose a date range that fits your question. Next, select the type of data you want. Hourly data is most helpful for understanding night conditions and wetness patterns. Daily summaries are better for weekly planning.
Degree Day Calculator
The Degree Day Calculator estimates accumulated degree days for a selected base temperature and location and often includes a near-term forecast. Degree days are most useful for insects and other pests whose development is strongly temperature-driven. Growers can use this tool to anticipate emergence and peak flight windows, time monitoring (traps/scouting), and compare season progress from one year to the next.
To use it, open the Degree Day Calculator and select a location. Then choose a date range. Common start dates include March 1, planting date, transplant date, or a pest-specific biofix date. Select the base temperature required for the pest or tool you are using (for example, base 40°F is common for maggot tools). Use the default calculation method unless you have a program standard you follow. Review the accumulated degree days, the accumulation graph, and the 5-day forecast, which is useful for planning when to intensify monitoring or schedule field operations. GDD is used in the forecasting models that will be discussed below.
Vegetable IPM forecasting models
The models use biology + weather risk + NEWA outputs to guide action.
Beet Cercospora Leaf Spot
Cercospora leaf spot is a polycyclic disease, meaning it can cycle repeatedly during the season when weather is favorable. The pathogen survives in infected residue and plant debris, so inoculum tends to build in fields with frequent susceptible hosts or poor residue management. Spores spread primarily by rain splash and wind-driven rain, but movement through wet foliage (equipment or workers) can also contribute.
Infection occurs when spores land on leaves and there is enough leaf wetness and favorable temperature for germination and penetration. As lesions develop, they produce more spores, allowing the disease to accelerate rapidly under continued favorable conditions. Crop susceptibility increases as canopy develops and leaves overlap, which traps humidity and prolongs wetness duration.
Outbreaks are favored by warm, humid weather with frequent showers, dew, fog, or overhead irrigation. Dry, breezy conditions slow disease progress but do not remove existing infections.
NEWA provides infection risk forecasts on multiple horizons: a near-term 2-day forecast, 14- and 21-day planning forecasts, and a season-long pressure view. Growers should use the 2-day forecast as an immediate trigger, if risk rises, scout within 24–48 hours, and if a protectant interval is nearing its end, apply protection before the risk spike rather than after. The 14–21 day views are most useful for planning sprayer logistics, scouting frequency, and fungicide rotation. The season-long view helps set overall intensity: high season risk supports a more disciplined, tighter approach, while low season risk paired with clean scouting supports avoiding early over-spraying.
In the field, start scouting with older leaves and the lower canopy, especially edges or zones that stay wet longer. Once lesions are established and weather remains favorable, progress can be fast.
Cabbage Maggot
Cabbage maggot is a soil-associated pest with multiple generations. It overwinters as pupae in the soil. Adults emerge, mate, and lay eggs near stems at the soil line. Larvae hatch and tunnel into roots, leading to wilting, stunting, plant collapse, and secondary rots. Seedlings and transplants are the most vulnerable; damage is most severe when larvae attack small root systems.
Because development is driven by temperature, degree days are a practical way to anticipate when adult activity and egg laying will occur. Cool springs can stretch adult activity, while warm periods compress development and speed egg hatch and larval feeding. Wet soils can increase plant stress and make symptoms worse; dry crusting may reduce egg survival in some conditions, but it is not reliable as a control tactic.
NEWA supports a base-40°F degree-day approach to help identify critical timing before larvae enter roots. Growers can use degree-day trends before planting to anticipate risk windows and prepare protection strategies such as barriers (row cover or netting) or planned treatments. At planting or transplanting, barriers (insect net) should be installed immediatelyand sealed at the edges. If insecticides are used, they must be timed so plants are protected before egg hatch and larval entry. Once larvae are inside roots, control is difficult.
During predicted peak risk, confirm activity with traps and scouting. Check for wilting or stunting, and pull suspect plants to inspect roots for tunneling and larvae. With cabbage maggot, timing is often more important than product selection; late applications frequently fail even with strong materials.
Onion Maggot
Onion maggot has a life cycle similar to cabbage maggot and is also strongly temperature-driven. It overwinters as pupae in soil. Adults emerge in spring, lay eggs near the base of plants, and larvae burrow into the bulb or neck region. Injury can cause stand loss, rot, and long-term storage issues. Seedlings and young plants are most vulnerable, but later infestations can still damage bulbs.
Weather influences are largely mediated through degree days: warm spells compress development and can bring peaks sooner, while cool conditions stretch the timing window. Wet conditions can worsen rot once feeding injury occurs.
NEWA provides daily and accumulated base-40°F degree days to anticipate emergence and peak risk windows. Growers use these outputs to start traps before flights intensify and tighten monitoring as degree days approach key windows. Protection and monitoring are often prioritized for early plantings, fields with a history of alliums, and areas near cull piles or volunteer alliums. Sanitation matters. Cull piles and volunteers increase local pressure and can overwhelm otherwise good timing.
Onion Diseases:
The onion diseases model combines weather-driven infection potential for several major foliar diseases. A practical way to use it is as a risk manager: when modeled risk rises, increase scouting and ensure your protection program does not have gaps.
- Botrytis leaf blight/blast is favored by cool to moderate temperatures and high humidity or leaf wetness. The pathogen produces spores that infect leaves and can cycle repeatedly when conditions stay favorable. Dense canopies that trap humidity increase risk. Infection is driven by wetness duration: prolonged dew, fog, and frequent light rains can matter more than heavy downpours. When model risk rises, tighten scouting and avoid letting protection lapse, especially in dense stands or where overhead irrigation extends wet periods.
- Onion downy mildew thrives under cool, wet, humid conditions and can spread rapidly during long stretches of high humidity and leaf wetness. Infection typically occurs during cool, humid nights, and sporulation often happens overnight. Early symptoms can be subtle, and outbreaks can appear to “jump” when favorable conditions persist. Long dew periods, fog, and low wind are major risk multipliers. When model risk increases, scout early in the day and focus on areas that stay wet; if disease is known regionally, treat rising risk more conservatively.
- Purple blotch is often linked to plant stress and injury (including thrips feeding, nutrient stress, and natural leaf aging). Spores are produced on infected tissue and spread by wind and rain. Infection requires leaf wetness, and lesion expansion is favored by warm, humid conditions. Alternating wet and dry periods can accelerate epidemics when wetness returns frequently. Management works best when paired with thrips control and practices that improve airflow and reduce prolonged wetness.
Across all three diseases, the decision-first use of NEWA is consistent: rising risk supports increased scouting and tighter protectant discipline, while low risk supports maintaining baseline scouting and avoiding interval tightening without field evidence. After irrigation or clustered rain events, revisit the model as foliar diseases respond quickly to short runs of favorable weather.
White mold in beans
White mold is driven by a well-defined soil survival and canopy microclimate cycle. The pathogen survives for years as sclerotia (hard resting bodies) in soil. Under cool, moist conditions, sclerotia can germinate to form apothecia, small “mushroom-like cups” that release airborne spores. Spores commonly land on flowers, colonize senescing petals, and then move into stems, producing white mycelium and new sclerotia. The most susceptible window is flowering through earlypod set, especially once the canopy closes.
Risk increases when canopy humidity remains high. Cool to moderate temperatures, frequent rain or overhead irrigation, and rapid canopy closure with low airflow strongly favor disease development. Because infection is closely tied to flowering and canopy conditions, timing matters and decisions can be costly.
NEWA provides a risk prediction based on 30-day weather trends combined with your field inputs such as canopy closure, irrigation status, and flowering stage. The most practical way to use this tool is as risk assessment. Use it to flag high-risk windows during early bloom, prioritize scouting in fields where canopy is closing and humidity is trapped, and adjust irrigation practices to avoid extended wetness during bloom when feasible. Field confirmation is still essential: validate risk by checking canopy conditions and scouting before committing to high-cost, timing-sensitive actions.
Want to learn more about NEWA as a tool for effective IPM?
Check out our upcoming NEWA Webinar Series!
Rosy Apple Aphid (Dysaphis plantaginea)
By: Evan Lentz, Assistant Extension Educator, Michael Fenton and Lauren Kurtz, Invasive Species Outreach Specialist
Hosts:
Apple and pear trees are the primary hosts for the Rosy Apple Aphid. The varieties ‘Cortland’, ‘Ida Red’, and ‘Golden Delicious’ are particularly susceptible. Alternate hosts for this pest include herbaceous plantains and docks.
Description and Life Cycle:
Rosy Apple Aphid (RAA), Dysaphis plantaginea (Passeri), is an early season pest of apples and pears. RAA overwinters as eggs laid on stems and bark of host trees. Pest emergence begins in early April as overwintering eggs begin to hatch. Newly hatched nymphs feed on developing buds and then migrate to developing leaves as they expand.


As first-generation nymphs reach reproductive maturity, females, called “stem mothers” give birth to live young through the process of parthenogenesis to form colonies (Figure 1). Nymphs remain close to the birthing mother and feed on leaf tissues. Toxins in the aphids’ saliva cause leaves to curl over at feeding sites, usually the underside of leaves (Figure 2). The curled leaves provide protection from natural predators, the environment, and insecticides, making early detection and control measures essential. Feeding sites will often harbor other aphid species although the RAA’s distinct rosy coloring should allow for differentiation.
The second generation of RAA usually appears 2 to 3 weeks post petal fall. Throughout the spring, populations swell as females continue to give birth, and aphids migrate from leaf to leaf. RAAs progress through five nymphal stages before reaching maturity within 2-3 weeks. As a result, RAA colonies produce and leave behind small masses of exoskeletons and a characteristic honeydew that promotes the growth of sooty mold.
In early summer, winged adult RAAs begin to develop and migrate off infested host trees to one of their herbaceous alternative hosts such as broadleaf plantain, narrowleaf plantain, and dock. On the alternate host, winged adults will reproduce asexually, producing additional generations of wingless adults.
In the fall, newly developed winged adults return to the primary host. Here, egg-laying females will mate with male aphids. RAA females will lay their fertilized eggs in hidden, creviced areas on the host tree, often around buds. However, recent research has suggested that RAAs may not need to migrate to alternate hosts and are instead able to complete their life cycle solely on their primary host.
Identification:
Eggs: RAA eggs are a creamy yellow color which slowly transitions to a shiny black in the winter. Egg masses can be found on the stems, at the base of spurs, and on buds throughout the hosts’ dormant season. Eggs are ½ mm long and football shaped. However, RAA eggs are not an effective way to identify or monitor this pest due to their likeness to other insect eggs.
Stem Mothers: Stem mothers are relatively large compared to nymphs and adults, with a rounded body and a thick white coating, making them appear grayer than the later rosy, purple-colored generations, and black antenna.
Nymphs: There are 5 nymphal stages which vary in size and color. Early nymphs are tan with a shiny, wet appearance and slowly transition to their characteristic rosy brown/purple color over time.
Adults: Wingless RAA adults are smaller and more purple than birthing females (Figure 3). Winged adults that migrate have dark brown/green bodies, a black head, a black thorax, and clear wings that develop one at a time. Adults are about 2.5 mm long.
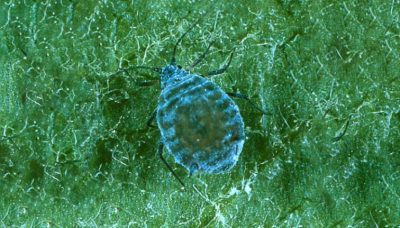

Damage:
Aphids utilize their piercing and sucking mouthparts to break into plant cells and tissues to extract sap and nutrients. This feeding can cause chlorosis and eventually necrosis. While feeding, RAA releases a toxin that causes deformation of leaves and developing fruit (Figure 2 & Figure 4). Damaged leaves often exhibit a contorted, puckered, or curled appearance. Damaged fruit typically exhibits puckering towards the calyx end. As the fruit develops, damaged tissues become lumpy, asymmetrical, and rigid (Figure 4). Additionally, aphid feeding sites provide entry points for other pathogens, effectively increasing the risk period past the bloom period of host trees. An additional secondary infection risk arises from the RAA colonies’ production of honeydew, which promotes the growth of sooty mold on developing fruit.
Monitoring:
Early scouting and identification of rosy apple aphids are integral to effective chemical control of the pest. Aphid eggs can be scouted for before or during early pink. Begin scouting on early season varieties at highest risk of economic damage occurring like Golden Delicious, Cortland, and Empire. By selecting 5 to 10 trees for every block, take at least 3 minutes and count each fruit spur showing curled leaves. If the average is more than one curled cluster per tree, an insecticide spray is economically and productively justifiable. Growers should be scouting closer to the inside and high parts of trees as this is where they are most likely to first appear. Looking for curled and deformed fruit is another major symptom that growers should be on the lookout for throughout the season.
Management:
The best chance growers have at controlling rosy apple aphid populations chemically is by applying sprays early in the season. Insecticidal soap at concentrations ranging between 1% and 2% or organophosphate insecticides are the most effective when it comes to control of early season RAA. Pesticides with a systemic mode of action are the most effective. Insecticides with the active ingredient spirotetramat are effective at restricting development and reproduction of RAA. This ingredient is slightly harmful or harmless to beneficial insects depending on the degree day it was sprayed and the population of the beneficials in the area. Remove secondary hosts around the area such as broadleaf plantain and doc as these harbor the aphids during the summer. Removing these resources disrupts their natural cycle and can prevent new winged adults from migrating back to apple trees.
References
Krawczyk G. 2023. Tree fruit insect pest – Rosy apple aphid. Penn State Extension. Accessed 5 Feb 2026. https://extension.psu.edu/tree-fruit-insect-pest-rosy-apple-aphid.
Piñero J. Aphid: Rosy apple aphid. New England Tree Fruit Management Guide. Accessed 5 Feb 2026. https://netreefruit.org/apples/insects/aphid-rosy-apple-aphid.
Rivera M, Basedow M, & van Zoeren J. 2022. Rosy apple aphid (Dysaphis plantaginea). Cornell Integrated Pest Management Program. Accessed 5 Feb 2026. https://cals.cornell.edu/integrated-pest-management/outreach-education/fact-sheets/rosy-apple-aphid-dysaphis-plantaginea.
What a Caterpillar Tunnel (and Climate Smart Microgrants) Can Do
By: Rebecca Toms, Food Systems Communications & Outreach Assistant, UConn Extension
In farming, sometimes one tool, one piece of infrastructure, or one infusion of amendments for your soil can make a big difference. For new and beginning farmers, this is especially true as they gather any resources to inch the needle closer to turning their new farm into a thriving business. The Solid Ground Climate Smart Micro-grants can help make that possible.
Since 2024, the Solid Ground Program at UConn Extension has offered new and beginning farmers who complete the Climate Smart Adaptation Strategies for Beginning Farmers course the opportunity to put their skills into practice by applying for a micro-grant. These grants (up to $2,400) give farmers the chance to implement what they’ve learned while giving their businesses a meaningful boost.
Gabby Rivera of Still Life Flower Farm in Canterbury has been growing flowers for a couple of years. With a caterpillar tunnel purchased through the 2025 micro-grant, she was able to improve the quality of her crops. “You can make so much more money if you can protect your plants,” Gabby said during a recent phone conversation. The tunnel now houses ranunculus—a famously finicky flower—and mums. Both crops are harvested during the shoulder seasons (early spring and fall), providing valuable income when the fields are otherwise quieter.

Ranunculus corms are expensive and growing them without protection often leads to inconsistent yields and lower quality blooms. At the same time, these vibrant, multi-layered flowers, are in high demand by florist and reliably sell at a good price point. When Gabby installed the tunnel, she knew ranunculus would be the priority crop. Since then, she has tripled her production.
 On the farm, she and her husband often talk about “before tunnel and after tunnel”—there’s simply no comparison. Growing under cover was one of the climate-smart practices highlighted in the course that backs up the experience Gabby is having on her farm. By stabilizing the growing environment, farmers like Gabby gain more control over their crops. Sudden heavy rains, strong winds, and unseasonable temperature drops—hallmarks of climate change—are far easier to manage when plants have protection from extreme weather.
On the farm, she and her husband often talk about “before tunnel and after tunnel”—there’s simply no comparison. Growing under cover was one of the climate-smart practices highlighted in the course that backs up the experience Gabby is having on her farm. By stabilizing the growing environment, farmers like Gabby gain more control over their crops. Sudden heavy rains, strong winds, and unseasonable temperature drops—hallmarks of climate change—are far easier to manage when plants have protection from extreme weather.
Gabby is one of over 50 farms to have received a microgrant through the program. Many farmers share similar stories of how these small investments, combined with the practices highlighted in the course, have made an impact on their operations.
To learn more about the micro-grants and the farms that have benefited, visit the Solid Ground website at solidground.extension.uconn.edu/climatemicrogrants and explore the practices farmers are adding to their operations.
The Climate Smart course will open again to new and beginning farmers at the end of 2026. We hope even more growers will take the online class and discover how adapting to a changing climate can strengthen both their farms and their bottom lines.
Growing in the City-Season Extension Without a High Tunnel
By: Jacqueline Kowalski, Associate Extension Educator, Urban Agriculture, University of Connecticut and Timothy McDermott DVM, Assistant Professor, Agriculture and Natural Resources, Ohio State University Extension, Franklin County
Seasonal high tunnels have become the norm for season extension. However, seasonal high tunnels have many disadvantages, and they may not be allowed on some urban farms. Many smaller-scale options/strategies are available. The goal of season extension is to identify, modify, and create microclimates to produce conditions favorable for crop production. Several strategies can be used in conjunction to create environmental conditions similar to those in high tunnels.
Variety selection
Some vegetable varieties are more cold-tolerant than others. Using cold-tolerant varieties can extend the production season both in the spring and fall. For example, growing the most cold-tolerant vegetable varieties during the coldest part of the year and then transitioning to those more tolerant of warm weather can extend the time certain vegetables, such as spinach, can be successfully grown.
Use of transplants vs. direct seeding
In general, using transplants that are several weeks old will produce a crop faster than direct seeding. Both seeds and transplants need to be planted when the soil is at the right temperature to successfully establish, and “the right temperature” varies by species. For additional information on the temperature required for specific vegetable seeds to germinate, read the results of a very interesting study conducted by Oregon State University on Soil temperature conditions for vegetable seed germination | OSU Extension Service. Season-extending fabric or plastic mulch can be used to raise soil temperatures. If the spring is excessively cool, it may take transplant longer to establish than normal even with the use of fabric or plastic mulch.
Site selection
Different areas on your farm may warm up earlier in the spring and cool down later in the fall. This could be due to slope, soil differences, or other factors. Taking advantage of microclimate differences on your farm can extend the season by using them to your advantage. South-facing slopes will warm up faster and stay warm longer during the season. Windbreaks may also moderate temperature.
Cloches
Cloches are an early form of season extension that is still used today. Originally, the term referred to a large glass jar placed over a tender plant to protect it from the cold. The term is now used interchangeably with small movable structures that can be vented in one way or another but heat up via capture of sunlight. Buckets, milk jugs, and plastic bottles can also be used to protect plants on a very small scale.


Plastic and biodegradable mulches
Plastic mulches are the standard in commercial vegetable production. During the day, soil temperatures can be 3-5 degrees higher in the soil profile. Some biodegradable mulches raise soil temperature, but others do not. Additionally, when using darker colored mulch, weed growth may be suppressed by blocking light from reaching the soil. Non-degradable plastic mulch may need additional management as it will not break down in the soil and should not be incorporated into the soil at the end of the season.
Covers
Multiple types of cover can be used to extend the season by creating microclimates. Each has benefits and drawbacks, individual costs, life span, and management requirements (such as removing and recovering the crop based on the weather and temperature). For the covers to work properly, the entire plant must be covered, and the material should be secured along the edges.
Row cover
Row cover (spun-bonded material) comes in different thicknesses or weights, which directly affect the amount of protection it provides. Heavier materials are more durable, provide a higher level of frost/freeze protection, but allow less light transmission. Lighter spun-bond material provides less frost protection, allows more light penetration, and is often less durable, but is commonly used as an insect barrier. All weights are permeable to water and air, so rainfall will reach plantings underneath, and heat buildup is not as problematic as with plastic. The heaviest-weighted row covers are often called frost blankets.


Plastic Film
Other materials that are used to cover low tunnels include plastic with slits or solid plastic. Solid plastic requires more management than row cover due to watering needs, potential rapid heat buildup, and condensation from moisture that falls on the growing crops, creating environmental conditions conducive to disease development. A common thickness used for plastic film is 6 mils.
Shade cloth
Not all methods of season extensions involve protection from cold. It is common to use season-extending methods to protect cool-weather-tolerant plants from excess sun or heat, allowing a longer harvest period (e.g., lettuce), or to protect fruits from sunscald (e.g., peppers and tomatoes). One method of warm weather protection involves using shade cloth. Shade cloth is a plastic mesh that allows both rain and air to pass through easily but reduces a certain percentage of sunlight. It is used to keep the area under the cloth cooler, benefiting and protecting crops. Shade cloth can be black, white, or grey. Shade cloth comes in different light transmissibility levels, measured as a percentage of sunlight they block and the degree of shade they provide.


Applications
Floating row covers
Floating row covers are when spun-bonded polyester or polypropylene is placed directly on top of growing crops, with the edges held down by bricks, sandbags, or boards. Enough slack is left in the material to allow the plant's upward growth. This method should not be used on crops with tender growing points. Any material that contacts plant foliage can serve as a point of transmission for cold, and plants may suffer frost damage at that point.
Low tunnels
Low tunnels are temporary structures that are easily installed, uninstalled, and moved with crop rotation. Low tunnels are usually 2-3 ft. high and cover one growing bed. Heavy-gauge wire, PVC pipe, and electrical conduit are among the materials that can be used for the tunnel's hoop section. Low tunnels can be covered with row cover (spun-bonded fabric), plastic film, or shade cloth, depending on the level of heating or cooling desired.
Caterpillar tunnels
Filling the niche between low tunnels and high tunnels, caterpillar tunnels offer flexibility for those not ready to invest in a high tunnel. Caterpillar tunnels are generally 5’ to 7’ at their highest point and offer several advantages over high tunnels. They are generally less expensive to construct, don’t need to be placed on flat ground, and are easily moved.
The ends of the hoops can be pushed directly into the soil or placed over the rebar that has been pounded in. Hoops can be spaced 6-8 feet apart, depending on the wind exposure. Hoops are covered with plastic during the coldest part of the season and can be replaced with other materials, such as shade cloth, at other times of the year. It is important to check with the municipality where you live to determine whether such structures are allowed and whether you need a permit to construct them.


Multiple Approaches
A multi-pronged approach, such as using climate-tolerant varieties, mulch, and low tunnels together, can amplify the effect of individual approaches. Layers can be added as needed for extreme cold protection.
It is important to note that structures such as low tunnels, caterpillar tunnels, etc. are vulnerable to wind and snow load. They need to be well-anchored, or they will blow off/away. They may also collapse under heavy snow, so it is critical to monitor the weather and remove the snow as needed.
Season-extension techniques can be successfully used on urban farms to increase production and profits. These techniques do take planning and, in some cases, capital investment, but in general, the effort will pay off.
References
ATTRA. 2005. Season Extension Techniques for Market Gardeners. Retrieved from: https://attra.ncat.org/product/season-extension-techniques-for-market-gardeners/#:~:text=Market%20gardeners%20can%20grow%20crops,markets%20and%20demand%20higher%20prices
Grubinger, V. 2015. High Tunnels and Other Season Extension Techniques. Retrieved from https://www.sare.org/resources/high-tunnels-and-other-season-extension-techniques/
Harington, J. F. 2013. Soil Temperature Conditions for Vegetable Seed Germination. Retrieved from: Soil temperature conditions for vegetable seed germination | OSU Extension Service
Upcoming Events
Farm Profitability Strategies for CT Growers

Tuesday, March 17th, 12-1pm
Join us for an informative webinar focused on practical strategies to improve farm profitability.
A panel of industry experts will share financial planning tools, real-world scaling insights, and approaches to product and market diversification that support sustainable farm business growth.
Speakers:
Chris Laughton (Farm Credit East)
Will O'Meara (Hungry Reaper Farm)
Bruce Gresczyk Jr. (Gresczyk Farms LLC)
Network for Environment and Weather Applications (NEWA)
Webinar Series
Wednesdays, March 18th, 25th, and April 1st
11am - 12:30pm
Have you heard of the NEWA network but never utilized its weather or pest forecasting tools?
Do you want another tool in your toolbox for thoughtfully managing pests on your farm?
Join us for an introductory webinar series on the functionality of the NEWA system. Our speakers will cover topics from degree-day calculations and blossom thinning to tracking insect pest emergence and disease pressures. This series will provide insights to both fruit and vegetable growers.
2026 CT Compost Conference

Thursday, March 19th
Wesleyan Universisty
Hosted by the Connecticut Compost Alliance (CCA), the CT Compost Conference brings together composters, farmers, educators, municipal leaders, policymakers, and sustainability advocates. Speakers from Connecticut and across the country will share innovative ideas and practical approaches to composting, soil health, and resilient systems.
Didi Pershouse is this year’s keynote speaker. Author of The Ecology of Care and Understanding Soil Health and Watershed Function. An internationally recognized educator and organizer, her work on soil health and watershed function is used in over 100 countries. She is co-founder of Rehydrate California and hosts The Wisdom Underground on Substack. Her keynote, “Growing Community and Climate Resilience through the Soil Sponge and Living Climate,” will explore how healthy soils strengthen ecosystems, communities, and climate resilience.
Additional Details
The conference is free for all to attend. Register for the conference.
Lunch is available for those who opt in, with a suggested $20 donation per person, which will be matched dollar-for-dollar at patronicity.com/compost2026. Attendees can also opt to bring their own lunch or venture out to the many dining options in Middletown, CT. This conference is made possible, in part, by a matching grant from Sustainable CT (sustainablect.org).
Quick Links
This work is supported by the Crop Protection and Pest Management Program [grant nos. 2021-70006-35582 and 2024-70006-43570] from the United States Department of Agriculture’s National Institute of Food and Agriculture. Any opinions, findings, conclusions, or recommendations expressed in this publication are those of the author(s) and should not be construed to represent any official USDA or U.S. Government determination or policy.
The University of Connecticut complies with all applicable federal and state laws regarding non-discrimination, equal opportunity and affirmative action, including the provision of reasonable accommodations for persons with disabilities. Extension program participants with disabilities may request reasonable accommodations to address limitations resulting from a disability. For more information, please contact the UConn Extension Civil Rights Liaison at extensioncivilrights@uconn.edu.
The information in this newsletter is for educational purposes. The recommendations contained are based on the best available knowledge at the time of publication. Any reference to commercial products, trade or brand names is for information only, and no endorsement or approval is intended. The Cooperative Extension System does not guarantee or warrant the standard of any product referenced or imply approval of the product to the exclusion of others which also may be available. All agrichemicals/pesticides listed are registered for suggested uses in accordance with federal and Connecticut state laws and regulations as of the date of printing. If the information does not agree with current labeling, follow the label instructions. The label is the law. Warning! Agrichemicals/pesticides are dangerous. Read and follow all instructions and safety precautions on labels. Carefully handle and store agrichemicals/pesticides in originally labeled containers, out of reach of children, pets and livestock. Dispose of empty containers immediately in a safe manner and place. Contact the Connecticut Department of Environmental Protection for current regulations. The user of this information assumes all risks for personal injury or property damage.






